With coal the most abundant of fossil fuels on our planet the search for clean coal power continues. A new 25-Kilowatt facility at Ohio State University reported the results of a week-long test process that if scaled could dramatically reduce pollutants such as nitrogen oxides and CO2 from coal-fired power plants. The next step involves ramping up to a 1 Megawatt facility.
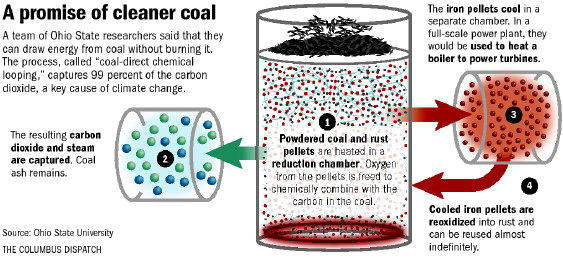
Today’s power plants using coal as the primary fuel combine air and a coal powder to create the energy to drive steam turbines. The process pollutes. Scrubbers capture the particulate matter in the outflow gases but do not reduce the CO2 and nitrogen oxide enough to impact greenhouse gas emissions. The pilot plant developed by Ohio State, however, uses a process called chemical looping which doesn’t expose the coal to air. Instead coal interacts with iron oxide producing CO2, iron and ferrous oxide in an artificial form called wustite (seen in an image below).
The burning of iron in air produces the energy that then gets converted into electricity. The process produces no nitrogen oxide, so no smog. And the chief byproduct of the process is pure CO2 which can be captured and sequestered.
Ohio State is also experimenting with a variation on this chemical looping process. This one involves gasifying the coal to create syngas, a combination of carbon monoxide (CO) and hydrogen. Iron oxide (rust) is introduced and reacts with the syngas to release oxygen and wustite. The oxygen combines with the CO to form CO2, and the hydrogen to form steam. The CO2 is streamed, captured and sequestered. The wustite then gets exposed to air and forms iron oxide (rust) and heat which then can be used to drive steam turbines. If the iron is exposed to steam it can separate out the hydrogen in the vapour and capture it as a fuel for fuel cells.
In another chemical looping test, calcium oxide from limestone is used to extract oxygen for use in burning coal. The process allows for the capture of pure CO2 while generating the energy needed to power steam turbines.
Although I have consistently ascribed oxymoron to the words “clean coal” it is encouraging to see that we may have answers that legitimately can allow us to continue to rely on coal as a primary energy source. Of course, that means retrofitting old coal powered plants or building new ones that used chemical looping technology. And it still doesn’t resolve the issue that we will have to sequester the CO2 byproduct produced by these power plants. That means finding a way to permanently trap the CO2 either deep in the Earth’s crust or in the deep ocean where it cannot emerge and further exacerbate the greenhouse effect.