One of my readers has challenged me to find evidence of ocean acidification on zooplankton and other sea life having pointed out that the studies I have cited included freshwater results or are purely in laboratories. That’s why I have chosen to highlight a story that appeared this week in the Los Angeles Times and in BBC Nature News.
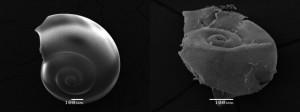
Both refer to a Nature Geoscience article published on November 25, 2012, entitled, Extensive dissolution of live pteropods in the Southern Ocean. The article is the published results of an in the wild study that looked at anthropogenic, that is human influenced, levels of CO2 and its impact on shelled marine creatures in the waters off Antarctica. The research data described was compiled from studying acidification levels and calcium carbonate levels in pteropods, marine snails, inhabiting the top 200 meters of a water column in the Southern Ocean. Data was collected in 2008 and concluded that these particular sea creatures were experiencing decalcification showing up as shell corrosion. The importance of studying pteropods is their place at the base of the ocean food chain and the potential impact their destruction could have on fish, seabird and whale populations.

The measurements looked at samples from cold seawater at depths of as much as 1,000 meters (3,300 feet) upwelling to the surface. Aragonite, a form of calcium carbonate that is found in pteropod shells, was measured. The researchers hypothesized that the combined effect of the natural upwelling of cold water with higher levels of CO2 was creating an acidity saturation horizon at depths from 200 meters to the surface where the pteropods thrive. That combination of the cold water and acidity appeared to be having a much higher corrosive impact on shelled creatures than anything observed in warmer waters.
You probably are asking yourselves what does cold water upwelling have to do with anthropogenic CO2 acidification? When deep cold ocean water rises to the surface and mixes with the more acidic shallow water, it cannot compensate for the cold water resulting in amplified acidification.
Who was involved and What did they Conclude?
Scientists from the British Antarctic Survey, the U.S. National Oceanic and Atmospheric Administration (NOAA), the Woods Hole Oceanographic Institution, and the university of East Anglia’s School of Environmental Sciences.
Their conclusions:
That ocean acidification from increased absorption of CO2 (correlated to human causes such as burning of fossil fuels) was the likely causal agent.
This is the first open ocean study looking at acidification and its impact on decalcification of a particular species in the ocean. Up until now all studies have been in laboratory settings. Described as a pilot project, the researchers intend to do further in the wild studies looking beyond pteropods to assess the impact of increased acidity on other ocean life.
Similar research is being conducted by scientists at the University of New South Wales in Australia who report that rising acid levels in the Southern Oceans will have a destructive impact on sea life within the next thirty years.
What ocean life is most vulnerable? Cold water coral communities, the nursery habitat for hundreds of commercial fish species, are probably most vulnerable, as are molluscs, starfish and sea urchins. In the community that constitutes phytoplankton, besides pteropods, we can expect similar decalcification impacts on foraminifera (single-celled amoeboid-like creatures with chalky shells) and coccolithophores (single-celled algae). And although shell corrosion does not necessarily mean the death of sea creatures that rely on calcium carbonate, any alteration in calcification rates that causes structural damage to organisms cannot be a good sign.

