In the December 29 issue of The Wall Street Journal an article authored by Ron Winslow describes a number of major advances in medicine aimed at transforming the way we treat and cure disease. One that immediately caught my attention deals with congenital heart disease (CHD) and a new surgical protocol to help children born with a rare heart lesion.
For those of you who know me, CHD is a subject near and dear to me because of my daughter. She was born with a rare heart lesion and has had multiple surgical corrections throughout her life. That’s one of the reasons I have been studying and writing about biomedicine for so many years.
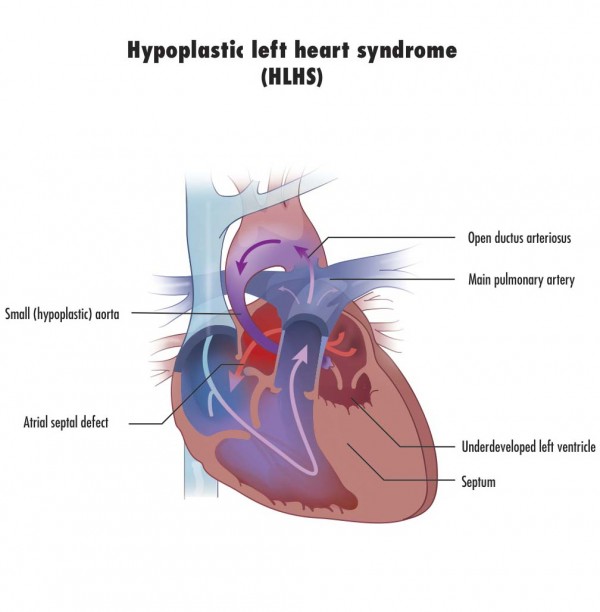
The new procedure comes from one of the hotbed facilities doing CHD research, Boston Children’s Hospital in Boston Massachusetts. The surgeons and cardiologists there have been working to find a way to let the body help itself in repairing the rare condition known as Hypoplastic Left Heart Syndrome (HLHS) seen in the image below.
Children born with this defect suffer from an underdeveloped, almost non-existent left-ventricle. Normally that venticle serves as the major pumping chamber of the heart pushing blood through the aorta to the rest of the body. But in HLHS children the chamber is malformed. When the child is in utero the condition is not life threatening. But once born HLHS without intervention is a death sentence. HLHS is very rare with maybe 1,000 children born in the United States and 100 in Canada each year. Early surgical intervention is the only chance these children have for surviving days let alone weeks, months or years.
When a HLHS baby is born they are immediately compromised. Early intervention attempts to keep the holes that exist in utero between the major blood vessels and the two upper chambers of the heart open. Normally these close within a few hours after birth. With the holes open there is at least some circulation of blood to the body’s major organs.
Surgical intervention prior to this new protocol has involved creating temporary fixes. The first is called the Norwood Procedure which turns the right ventricle used to pump blood to the lungs into a single pumping chamber pumping blood to both the lungs and the rest of the body. Using the right ventricle this way has drawbacks. The right side of the heart is less muscular than the left because the circulatory demands on it are far less than the left. In turning it into a dual-purpose pumping chamber the muscle gets highly stressed and eventually fails. That’s because there isn’t enough muscle to do both jobs.
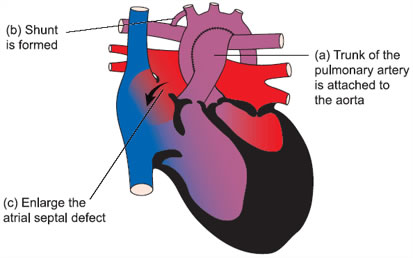
If a child survives for a period of time after a Norwood then other procedures can be done to try and create as close to a permanent fix as possible. But ultimately the preferred treatment for any HLHS child is to find a donor heart. Transplantation comes with its own challenges including donor availability and rejection management and therefore is considered a last resort. Surgeons would far prefer to use a body’s original parts whenever possible to affect a cure. That’s why finding a way for the body to self heal is considered ideal.
So the team at Boston Children’s set out to do just that, find a way to stimulate the left ventricle to grow from its hypoplastic state to a near normal size. Their newly developed surgical protocol is a technique that involves directing blood not away from but to the underdeveloped left ventricle and by doing this triggering the ventricle to grow so that it can eventually assume its normal function.The doctors who have pioneered this technique recently reported preliminary results in the Journal of the American College of Cardiology. They selected 34 patients for the new procedure and reported that 12 of them now have two working ventricles.
What’s involved with the protocol? Multiple surgical interventions. The first procedure alters the circulation of blood in and around the heart to stimulate left ventricle growth. Secondary procedures follow to repair or replace malformed or occluded heart valves, close off any holes between the pumping chambers, and reduce the amount of inelastic tissue in and surrounding the heart so that it can pump more efficiently.
Considering past results, even with multiple surgical procedures, this new protocol should give HLHS children a fighting chance at a full life.














Interestingly innovative article but I am shocked and appalled that this sentence was written,
“But ultimately the preferred treatment for any HLHS child is to find a donor heart.” Actually the preferred treatment is to try and have the child remain with their own heart for as long as possible. The author talks about transplantation like it’s just easier to handle. It’s very obvious that this author has never met a patient or parent of a patient who has been through an organ transplant. Most, not all, transplanted organs last between 8-10 years and then need to replaced.
I’m hoping with new procedures and biomedicine, HLHS can someday be a distant memory. In the meantime, organ transplant should not be the goal unless everything else has been exhausted and this all one is left with.
God Bless you. Our grandson is HLHS and is almost 16. He had the 3 Surgeries within his first 2 years. He is amazing and awesome and has a heart of Gold. I pray that he may have an opportunity for some kind biomedicine cure or procedure comes along for him. Sometimes he struggles but keeps going on. The Heart Transplant like you said is really a Temporary fix and comes with many complications. Prayers for you and your health.
My thoughts are with you for your grandson and family. Our daughter with 3 heart surgeries and arterial dissections and repairs is now 33.
In stating preference, it is not the opinion of the writer, but that of the majority of cardiac surgeons. It was the same when our daughter was born with CHD. We heard it from the medical team looking after her. Fortunately there were others who suggested a very different protocol.