Stem cells have become a part of the medical lexicon in the second decade of the 21st century. Where are we now with this promising scientific research and where will be going over the next 20 years. That is the subject introduced in this blog.
Stem Cells
These life-changing undifferentiated cells are generated and harvested from 3 sources.
- Embryonic Stem Cells
- Somatic or Adult Stem Cells
- Induced Pluripotent Stem Cells
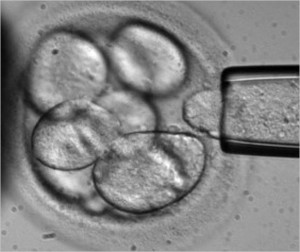
A human embryo in its earliest stages is called a blastocyst. It consists of two types of cells, an outer wall that becomes placental tissue and an inner wall of cells that eventually differentiate into all the tissues of the body. It is these latter undifferentiated cells that are potential tools for fighting disease and healing damaged tissue. This is a new field of reparative medicine done at the cellular level.
Stem cells can be reprogrammed. They have unique regenerative powers. Studying their growth is helping scientists to understand what causes birth defects. Currently researchers study stem cells for their potential to deal with a range of human diseases and problems including: stroke and traumatic brain injury, Alzheimer’s, Parkinson’s, blindness, deafness, traumatic wounds, ALS (amyotrophic lateral sclerosis), muscular dystrophy, diabetes, Crohns, cirrhosis of the liver, spinal cord injury, rheumatoid and osteoarthritis, heart disease, heart lesion repair after a heart attack, and many cancers. Researchers are even using stem cells to design and improve pharmaceuticals to target specific diseases and medical conditions.
Embryonic Stem Cells
We have experimented with embryonic stem cells since 1981 when scientists derived stem cells from the embryos of mice. By 1998 we discovered how to do this with human embryos. Only embryos discarded from in vitro fertilization procedures were used but nonetheless controversy soon erupted from religious groups that saw using these embryos as the equivalent of murder.
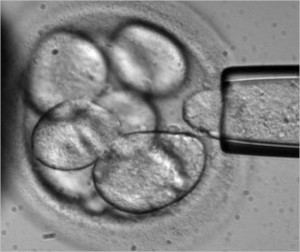
Embryonic stem cells can be cultured in a laboratory to divide while retaining their undifferentiated state. Called Pluripotent embryonic stem cells, they form stem cell lines which can be frozen and sent to laboratories and medical facilities for further research and testing.
In studying these cells researchers learn about human development and the complex processes involved when undifferentiated cells begin to differentiate to form various body tissues. This will help in understanding the mechanism that can lead to abnormal cell division and differentiation resulting in cancer and birth defects.
Somatic or Adult Stem Cells
Less controversial than embryonic, somatic (meaning cells not harvested from embryos) stem cells consist of undifferentiated cells found within body tissue that mostly contains differentiated cells. Somatic stem cells exist in organs and tissue in a caretaker role, providing maintenance and repair. We have been working with a form of somatic stem cells for some time. Probably the most well-known use is the procedure known as bone marrow transplantation used to treat leukemia, where hematopoietic stem cells are harvested from a donor and transplanted into a recipient.
Somatic stem cells have already been used to show their potential to create an endless supply of blood for transfusion allowing patients to bank cells that can be generated as needed eliminating the need for blood donors.
As researchers discover somatic stem cells in many more organs and body tissue their usefulness for other types of transplants may lead to cures for Parkinson’s, Alzheimer’s, heart disease, and many other intractable medical conditions.
Induced Pluripotent Stem Cells
Known in short form as iPSCs, these are adult tissue and organ cells that have been genetically reprogrammed to assume an undifferentiated embryonic stem-cell state. First “invented” and identified through the genetic manipulation of mouse cells in 2006, by 2007 human iPSCs had been harvested and cultured in laboratories.
Currently iPSCs are used for drug testing and modeling of diseases. Genetically altering an adult cell is no simple task. Using viruses as delivery agents, altered genetic materials get introduced into the cell. Unfortunately in some animal studies the viruses when introduced have caused cancer. We are not, therefore, ready to deploy iPSCs for therapeutic purposes in human studies. Researchers are exploring non-viral methods of delivering new instructions. Once they have mastered this the potential for therapies is enormous.
iPSCs can overcome tissue rejection. They can be used to repair and grow healthy tissue. This is the science of cell-based therapy and has the potential of replacing organ and tissue transplants. iPSCs can be created from a human source, cultured and directed to differentiate into specific cell types to treat diseases such as diabetes creating healthy insulin-generating cells in the pancreas, or repairing brains suffering from Alzheimer’s and Parkinson’s, or grow new heart muscle and blood vessels to cure heart disease, or repair immune systems that lead to osteo and rheumatoid arthritis. iPSCs have the potential to repair traumatic injuries such as spinal cord injury, or stroke, and regrow healthy skin to repair burns.
Stem Cell Therapy in the Medical Mainstream
How far are we away from seeing stem cell therapy in the medical mainstream? Today stem cells are being used to treat a small number of diseases and largely in clinical trial only. The most common use is blood stem cell transplantation for treating cancer. The PISCES study (Pilot Investigation of Stem Cells in Stroke), a fully regulated clinical trial of neural stem cell therapy for stroke patients started in 2010 in the U.K. In the United States clinical trials have started using embryonic stem cells differentiated into retinal cells to treat macular degeneration.
Soon these studies will be joined by many more. But first cell-based therapies need to move out of research laboratories. into many more clinical trials and then produce documented and consistent results to show that manipulated stem cells can be differentiated for transplantation consistently. That means creating enough cell volume before differentiation and controlling differentiation to create desired tissue types that match individual patients. That means ensuring that the tissues created from stem cells function the way they are supposed to for a lifetime and that the human host doesn’t experience rejection or develop any diseases resulting from transplanted cells.
We should be there if not within this decade then by 2030.